Pyrolysis of Plastic
Pyrolysis is a process of thermal degradation of a material in the absence of oxygen. Plastic is continuously treated in a cylindrical chamber and the pyrolytic gases condensed in a specially-designed condenser system to yield a hydrocarbon distillate comprising straight and branched chain aliphatics, cyclic aliphatics and aromatic hydrocarbons. The resulting mixture is essentially equivalent to petroleum distillate. The plastic is pyrolised at 370ºC-420ºC and the pyrolysis gases are condensed and liquid separated using fractional distillation to produce the liquid fuel products.
The essential steps in the pyrolysis of plastics involve:
- evenly heating the plastic to a narrow temperature range without excessive temperature variations
- purging oxygen from pyrolysis chamber,
- managing the carbonaceous char by-product before it acts as a thermal insulator and lowers the heat transfer to the plastic
- careful condensation and fractionation of the pyrolysis vapors to produce distillate of good quality and consistency
Flow Diagrams
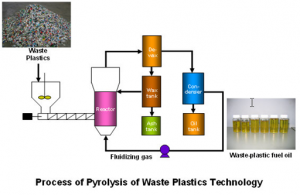
(Source: http://amudu-gowripalan.blogspot.com/2014/10/production-of-fuel-and-diesel-from-non.html)
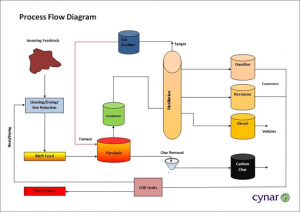
(Source: http://www.pacebutler.com/blog/plastic-to-fuel-recycling-gaining-traction-in-the-us/)
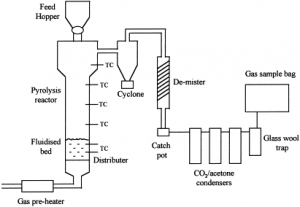
(Source: http://ars.els-cdn.com/content/image/1-s2.0-S016523709900011X-gr1.gif)
Equipment
Feed Hopper
Reactor
Fluidized bed
Distributer
Cyclone
Catch pot
Condenser
Videos
Hong Kong Company Converts Plastic to Fuel
References
- http://www.youtube.com/watch?v=BeccQTyGVX0
- http://www.cynarplc.com/cynar_technology.asp#Pyrolysis
- http://ars.els-cdn.com/content/image/1-s2.0-S016523709900011X-gr1.gif
- http://www.cynarplc.com/images/ProcessFlowDiagram.jpg
- http://www.itri.org.tw/eng/UpFile/_userfiles/image/Fuel%20Oil%20Produced%20from%20Pyrolysis%20of%20Waste%20Plastics.jpg