Liquefaction of Algae
If biomass were grown for energy to an amount equal to that consumed during their any given production period, there would be no net buildup of CO2 in the atmosphere (Gao and Mickinley 1994 ). Microalgae are particularly promising biomass species because of the high growth rate and high CO2 fixation ability compared to plants (Tsukahara and Sawayama 2005 ).
Algae provide many potential routes for the conversion into biofuels, including hydrothermal liquefaction. During hydrothermal liquefaction, high moisture biomass is subjected to elevated temperatures (250-350 deg C) and pressures (10-20 MPa) in order to break down and reform the chemical building blocks into bio-crude oil. At these temperatures and pressures, water becomes a highly reactive medium promoting the breakdown and cleavage of chemical bonds, allowing for the reformation of biological molecules. The conversion mimics the natural geological processes which produced our current fossil fuel reserves and allows for the conversion of a wide range of feedstocks.
Water is also beneficial as a reaction medium since the newly formed bio-crude oil self-separates after conversion. The aqueous medium also eliminates the need dry the incoming feedstock, bypassing resource and energy intensive preprocessing steps. Depending on the feedstock, the resulting bio-crude oil can have a heating value comparable to bunker crude oil (30-40 MJ/kg) and can be burned in boilers or upgraded and refined into higher value fuel or chemical compounds.
Flow Diagrams
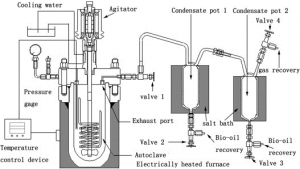
(Source: http://ars.els-cdn.com/content/image/1-s2.0-S0960852411016038-gr1.jpg)
Equipment
Temperature control device
Agitator pressure gauge
Electrically heated furnace
Videos
SURE 2012: Algae Biofuel: Hydrothermal Liquefaction of Microalgae
References
- http://www.youtube.com/watch?v=Ktii9ooWPuU
- http://algae.illinois.edu/Projects/Hydrothermal.html
- http://age-web.age.uiuc.edu/bee/research/IntroHTL.pdf
- http://ars.els-cdn.com/content/image/1-s2.0-S0960852411016038-gr1.jpg